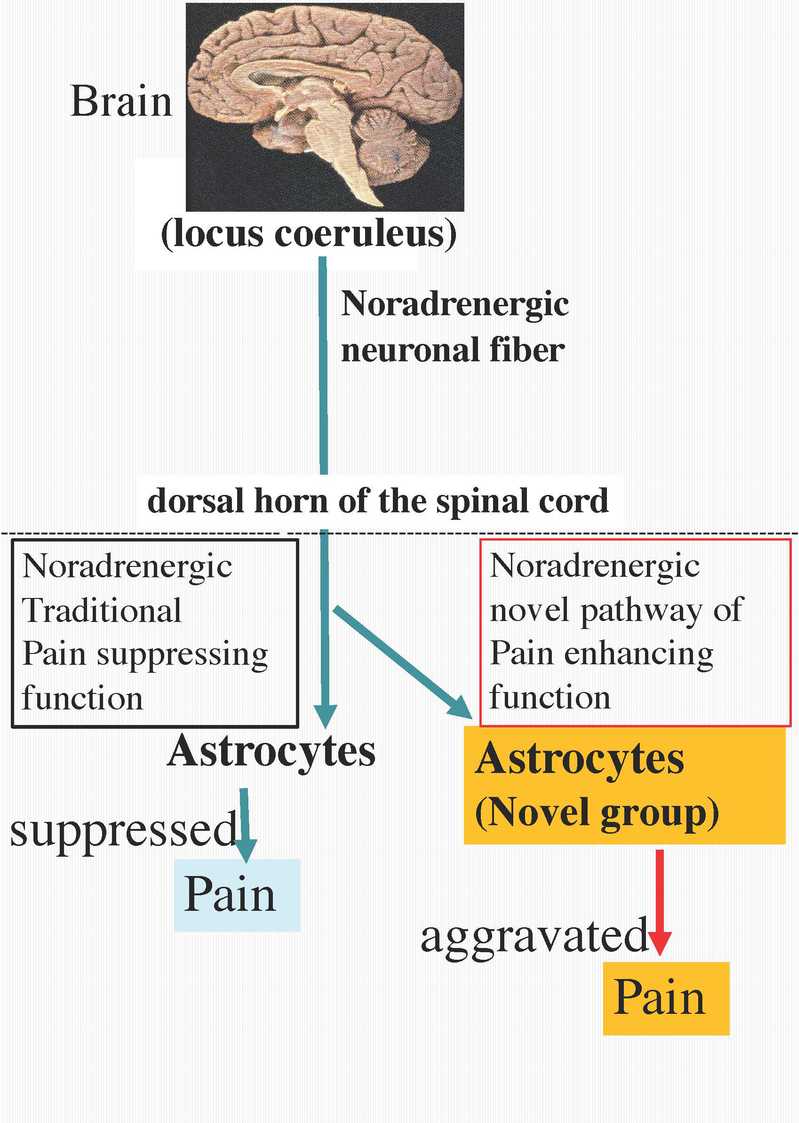
Professor Katsuhiko Mikoshiba from the School of Life Science and Technology (SLST) and SIAIS, in collaboration with Professor Makoto Tsuda’s group in Kyushu University, have published research findings in Nature Neuroscience describing a novel type of astrocyte involved in pain regulation: Noradrenergic stimulation aggravates pain through the novel astrocytes, opposite to the classical regulation, suggesting a possible new modality for pain therapeutics by suppressing the novel astrocytes, while traditional “duloxetine” suppresses the pain through noradrenergic pain suppressing function.
Pain is an important sensation for protecting ourselves. Astrocytes play a role in nociception: the role of nociceptors is considered a physiological self-protection mechanism to avoid external physical contact to the painful and neighboring areas. In humans and rodents, intense activation of nociceptors by capsaicin causes mechanical hypersensitivity. The noradrenergic pathway from the brain (Locus caeruleus) to the dorsal horn of spinal cord is considered to inhibit the pain sensation. In addition, astrogliogenic mechanical hypersensitivity requires Ca2+ signals in these cells. IP3 receptor 2, a Ca2+ releasing channel, is highly involved in nociception. In fact, the loss of IP3 receptor 2, which is crucial for astrocytic Ca2+ responses, prevents the mechanical hypersensitivities caused by chemogenetic astrocyte stimulation and intraplantar capsaicin. Intrathecal injection of noradrenaline produces anti-nociceptive effects. Therefore, adrenergic inhibition is considered to be one of the therapeutic tools for nociception.
In this study the researchers identified a novel population of astrocytes located in the particular astrocytes group (at superficial laminae of the spinal dorsal horn (SDH)) in adults that is genetically defined by Hes5. SDH is the relay point of sensory signal to the brain. In vivo imaging revealed that noxious stimulation by intraplantar capsaicin injection activated SDH astrocytes via adrenoceptors through descending noradrenergic signaling from the locus coeruleus in the brain. Intrathecal noradrenaline induced mechanical pain hypersensitivity via adrenoreceptors in Hes5+ astrocytes, and chemogenetic stimulation of Hes5+ SDH astrocytes was sufficient to produce the hypersensitivity. Furthermore, capsaicin-induced mechanical hypersensitivity was prevented by the inhibition of noradrenergic signaling onto Hes5+ astrocytes. Moreover, in a model of chronic pain, adrenoreceptors in Hes5+ astrocytes were critical regulators for determining an analgesic effect of “duloxetine”. Mikoshiba and his collaborators’ findings identified a novel astrocyte population that gates descending noradrenergic control of mechanosensory behavior.
Link: https://doi.org/10.1038/s41593-020-00713-4