Lab of Stem Cells and Epigenetics
Research Interests
Epigenetics in Development and Disease
DNA methylation and histone modifications are key epigenetic factors involved in gene regulation and cell physiology. Dysregulation of DNA methylation and histone modifications has been associated with several human diseases, including Fragile-X, ICF (immunodeficiency, centromere instability, and facial anomaly), Overgrowth, Dwarfism, and Rett Syndromes.
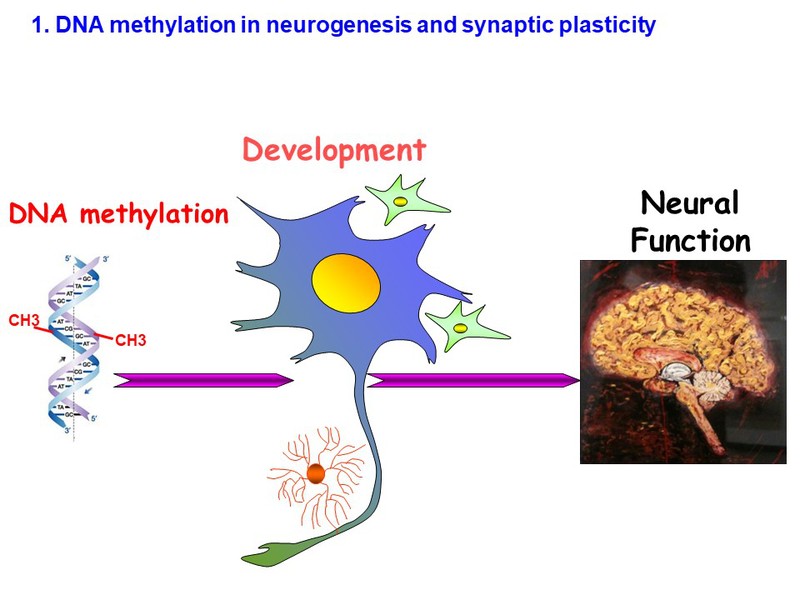
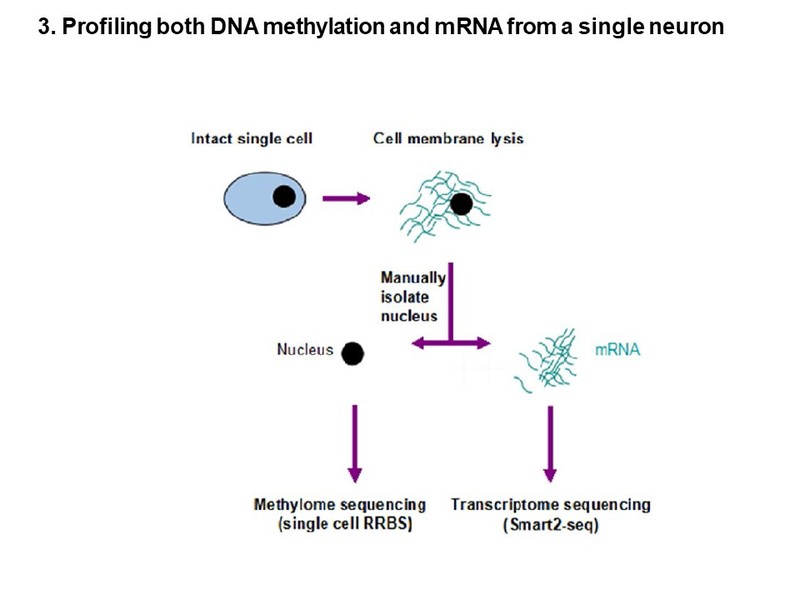
To study the function of DNA methylation in development, we have previously used the Cre/loxP system to knockout DNA methyltransferases (Dnmts) tissue-specifically. More recently, we applied the CRISPR-cas9 tool to mutate Dnmts at a single nucleotide resolution to model human genetic mutations in transgenic mice. By single-cell sequencing technique, we are currently defining the molecular mechanism by which DNA methylation alters neuronal gene expression, cell survival, and neuronal function.
Stem cells and Translational Medicine
The recent breakthrough of stem cell differentiation and reprogramming provides us with a unique opportunity to model human genetic disorders in stem cells. One of the focuses in our lab is to establish human iPSCs carrying genetic mutations leading to human diseases such as Overgrowth, Dwarfism, and ICF Syndromes that are caused by DNMT3A and DNMT3B point mutations. These human disease-specific iPSCs will be powerful model systems to dissect molecular and epigenetic mechanisms underlying these human diseases.
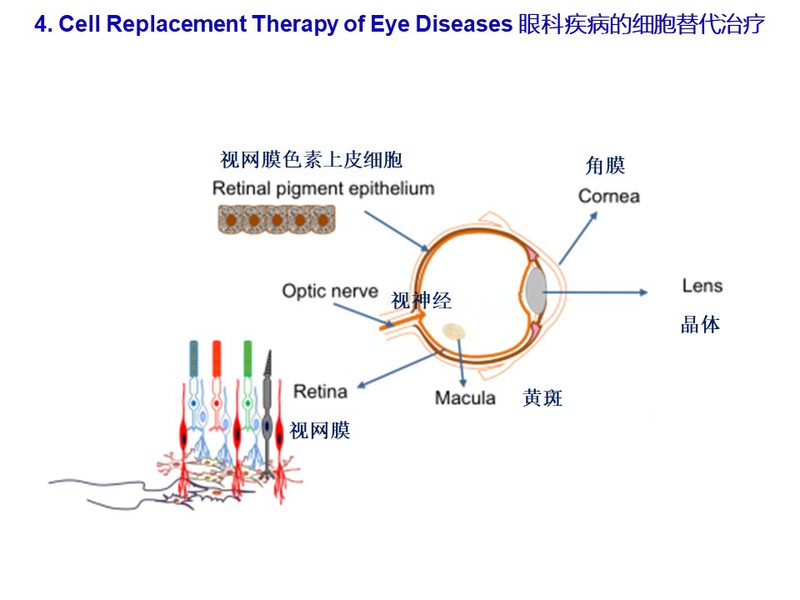
We also study stem cell differentiation towards specific cell lineages including retinal pigment epithelial cells, corneal endothelial cells, retinal ganglion cells via directed differentiations in 3D-retinoids. By cell transplantation in animal models of retina degeneration, we will test the efficacy of transplanted retinal and corneal cells in the rescue of visual acuity of animal models. Our ultimate goal is to translate our cell transplantation method to treat human patients with blinding diseases such as age-related macular degeneration and retinitis pigmentosa.
Developing advanced biomaterial platforms for cancer research
Conventional 2D cell cultures and in vivo rodent models have been used to assess the effects of drugs on tumor cell growth for many decades. However, they often fail to faithfully recapitulate the native human tissue-specific microenvironment. It remains challenging to design ideal materials that are flexible, biocompatible and capable to be applied for high-throughput assessment and screening.
We have developed a series of multifunctional polymers and injectable hydrogel systems via controlled living polymerization and bio-conjugation methods. These polymers can rapidly form stable hydrogels with modified bio-macromolecules via chemical or photo-cross-linking approaches. Two-photon high-resolution 3D printing and microfabrication techniques were utilized to construct controllable micro-patterns within the hydrogels. We are also developing an organoid-on-a-chip system to study dynamic interactions between tumor cells and material microenvironments. Aim to understand the biological mechanism of cell-cell and cell-ECM interactions during cancer progression, and to develop advanced in-vitro 3D tumor model for cancer research and drug screening.
Research Projects